Investigative journalism
-

Adults in the Student Suicide Case
Are Reporters Affiliated With Organizations Journalists or Just Employees of Organizations? / “Freedom of Press Trial,” The Second Oral Pleading
2024.01.30 14:38 Nanami Nakagawa
-
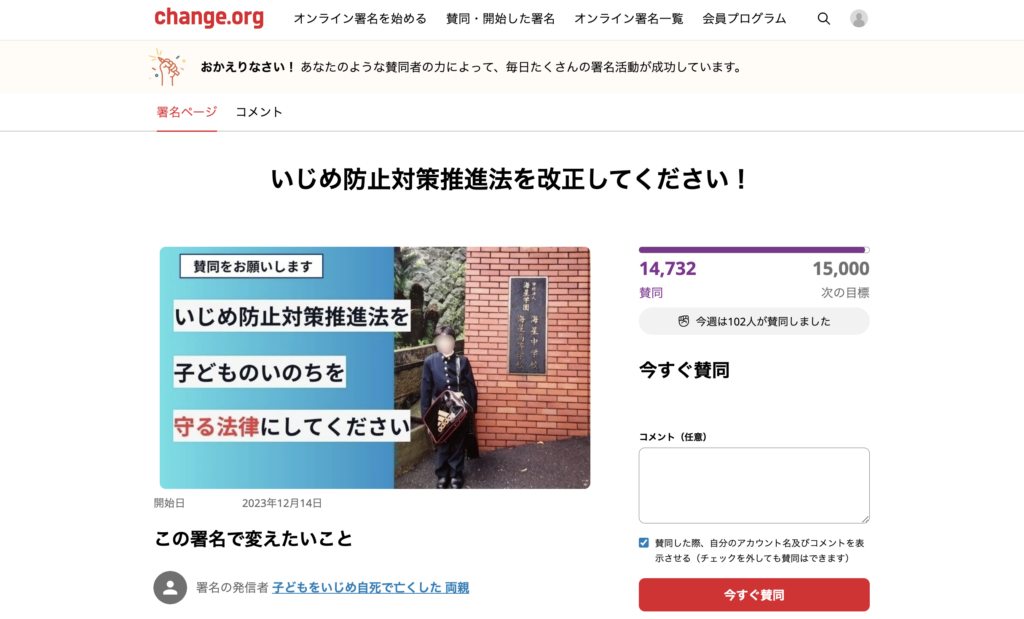
Adults in the Student Suicide Case
“How Many Children Have Committed Suicide As a Result of Bullying in The Last Ten Years?” / Bereaved Family Begins Collecting Signatures Calling For Law Reform
2024.01.25 10:36 Nanami Nakagawa
-

Adults in the Student Suicide Case
Questions The Judge Raised About Kyodo News / The First Oral Pleading of A Lawsuit Seeking Freedom of The Press
2024.01.19 12:08 Nanami Nakagawa
Announcements View all
- 2021.03.09 15:11
- Waseda Chronicle reborn as Tansa

 Newsletter signup
Newsletter signup












